Water Testing and Analysis
Quality Control of Pharmaceutical Water
What is Water Testing and Analysis?
Purified Water is the number one raw material used in pharmaceutical manufacturing. It is used at almost every stage of the manufacturing process from washing and cleaning to the primary ingredient in a final non-intravenous product formulation. It is produced through a multi-stage purification process usually including ion exchange and reverse osmosis. Water for injection is a further purification of pharmaceutical Purified Water via distillation, this distillation step removes bacterial endotoxins from the water. Due to the criticality of water and water systems, it is a regulatory requirement to perform routine chemical quality assessments.
Why do we do it?
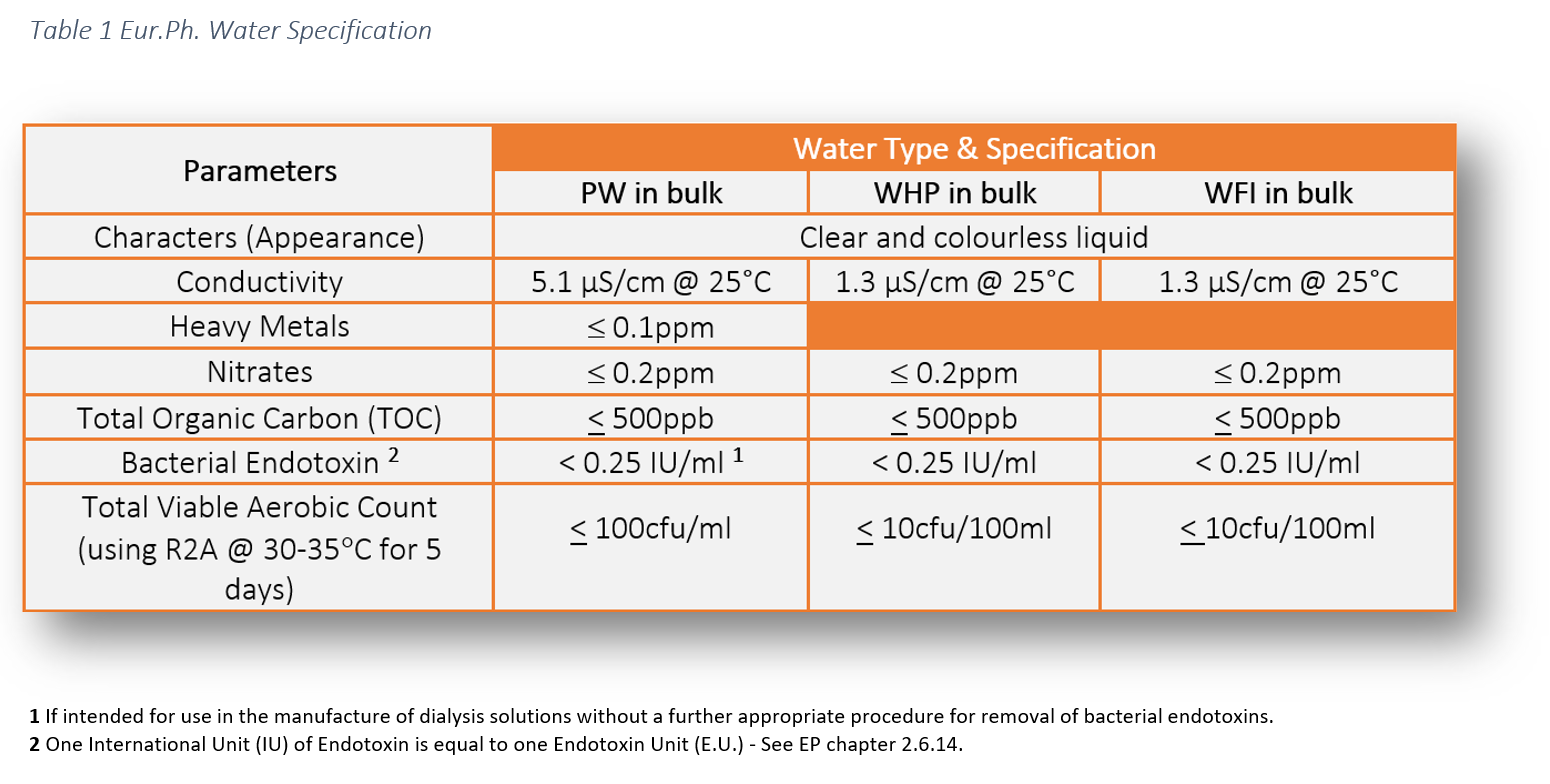
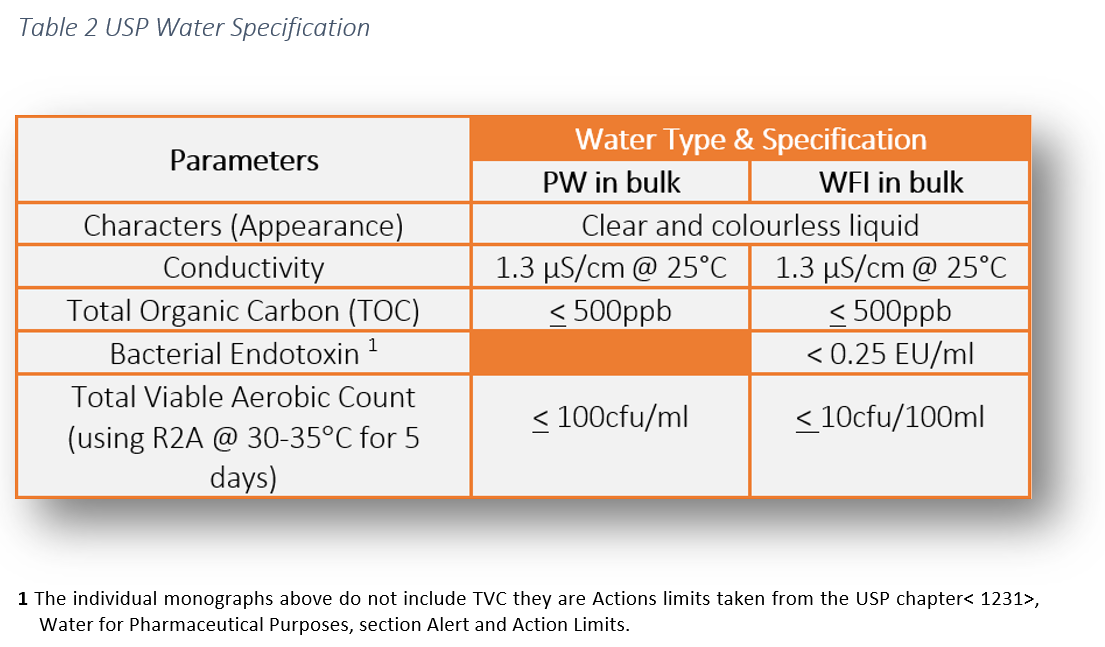
The legal requirement to comply with pharmaceutical water specifications is defined in the pharmacopoeial monographs. These water specifications can be found in the following pharmacopoeias: USP, Ph Eur and JP. This literature establishes the minimum quality standards within the industry which are enforceable by the regulators. As manufacturers and services providers we have a responsibility to ensure the highest level of operational control to ensure product safety for patients.
How do we assure compliance?
At Honeyman we are able to provide the full suite of cGMP microbiological and chemical testing for Purified Water and Water for injection to ensure compliance with the following:
- European Pharmacopoeia (Ph. Eur.)
- United States Pharmacopoeia (USP)
- Harmonized tests covering both Ph. Eur and USP (EP/USP)
- Japanese Pharmacopoeia (JP)
- HTM2030
- In house specifications
We cover the chemical tests detailed by major pharmacopoeias including:
- Conductivity
- pH
- Nitrates
- Total Organic Carbon
- Heavy metals
In addition we offer the following tests to suit client requirements:
- Calcium and Magnesium
- Chloride
- Arsenic
- Iron
- Heavy Metals
- Sulphate
- Potassium Permanganate
- Oxidisable Substances
- Total dissolved solids
- Manganese
- Silicates
- Phosphate
- Ammonium
Our Customers:
























